Anti-inflammatory Activity of Tectona grandis Linn. F. Extract Against TNF-α using In Vivo and Silico Methods
Downloads
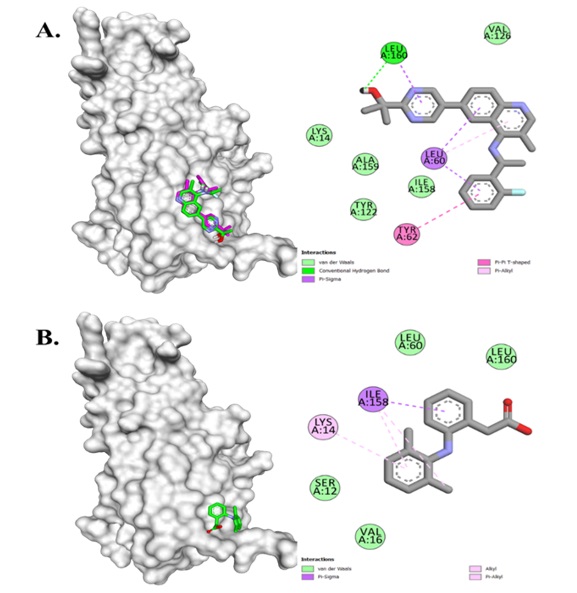
Tumor necrosis factor-alpha (TNF-α), a cytokine involved in inflammation, plays a critical role in inflammatory processes, particularly in synovial cells. Therefore, TNF-α represents a promising target for anti-inflammatory interventions. This study investigates the anti-inflammatory potential of Tectona grandis leaf extract by evaluating its ability to inhibit TNF-α activity. To identify bioactive compounds from the extract that may interact with the TNF-α receptor, molecular docking simulations were employed. In vivo experiments were conducted using different doses of the extract (100, 200, and 300 mg/kg body weight), and TNF-α levels were quantified to assess the anti-inflammatory effect. At a dose of 300 mg/kg, TNF-α levels were significantly reduced to 0.358 μg/L, indicating superior anti-inflammatory activity compared to the lower doses (0.539 μg/L at 100 mg/kg and 0.433 μg/L at 200 mg/kg). Molecular docking simulations revealed two bioactive compounds, Phaeophorbid A and (132S, 17S, 18S)-132-Hydroxy-20-Chloro-Ethylpheophorbide A, which demonstrated strong binding affinities (-5.4 kcal/mol and -5.2 kcal/mol, respectively) and interacted with the TNF-α receptor through hydrogen bonds and hydrophobic interactions. These results underscore the potential of Tectona grandis as a source of novel anti-inflammatory agents, suggesting its value in the development of therapeutic strategies targeting TNF-α in inflammation-related disorders.
Downloads
Marshall JS, Warrington R, Watson W, Kim HL. An introduction to immunology and immunopathology. Allergy, Asthma Clin Immunol. 2018;14(2):6–14. DOI:https://doi.org/10.1186/s13223-018-0278-1
Netea MG, Schlitzer A, Placek K, Joosten LAB, Schultze JL. Innate and Adaptive Immune Memory: an Evolutionary Continuum in the Host’s Response to Pathogens. Cell Host Microbe. 2019;25(1):13–26. DOI:10.1016/j.chom.2018.12.006
Rosenblum MD, Gratz IK, Paw JS, Abbas AK. Treating human autoimmunity: current practice and future prospects. Sci Transl Med. 2012 Mar;4(125):1–20. DOI:10.1126/scitranslmed.3003504
Chen L, Deng H, Cui H, Fang J, Zuo Z, Deng J, et al. Inflammatory responses and inflammation-associated diseases in organs. Oncotarget. 2017;9(6):7204–18. DOI:10.18632/oncotarget.23208
Jang D, Lee A-H, Shin H-Y, Song H-R, Park J-H, Kang T-B, et al. The Role of Tumor Necrosis Factor Alpha (TNF-α) in Autoimmune Disease and Current TNF-α Inhibitors in Therapeutics. Int J Mol Sci. 2021;22(5):1–16. DOI:https://doi.org/10.3390/ijms22052719
Asdaq SMB, Nayeem N, Abida, Alam MT, Alaqel SI, Imran M, et al. Tectona grandis L.f: A comprehensive review on its patents, chemical constituents, and biological activities. Saudi J Biol Sci. 2022;29(3):1456–64. DOI:https://doi.org/10.1016/j.sjbs.2021.11.026
Rudin NA, Hulaimah A, Rahmawati R. The Potential Utilization of Young Teak Leaves (Tectona grandis Linn.f.) as Basic Ingredients on Producing Anti-cancer Herbal Tea. J Pendidik Biol Undiksha. 2022;9(1):10–23.
Varma SB, Giri SP. Study of wound healing activity of Tectona grandis Linn. leaf extract on rats. Anc Sci Life. 2013 Apr;32(4):241–244. DOI:10.4103/0257-7941.131984
Diallo A, Gbeassor M, Vovor A, Eklu-Gadegbeku K, Aklikokou K, Agbonon A, et al. Effect of Tectona grandis on phenylhydrazine-induced anaemia in rats. Fitoterapia. 2008;79(5):332–336. DOI:https://doi.org/10.1016/j.fitote.2008.02.005
Tungmunnithum D, Thongboonyou A, Pholboon A, Yangsabai A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines. 2018 Aug;5(3):1–16. DOI:https://doi.org/10.3390/medicines5030093
Asif M. In Vivo Analgesic And Antiinflammatory Effects Of Tectona Grandis Linn. Stem Bark Extracts. 2012;9(1):1–11.
Parawansah, Nuralifah, Ruslin, Nurramadhani Armada Sida, Fadhliyah Malik A. Effects of Tectona grandis L . Extract in Diabetic Rats on Nitric Oxide and Malondialdehyde Levels. Pakistan J Biol Sci. 2023;26(4):177–184. DOI:10.3923/pjbs.2023.177.184
Budianto P, Suroto S, Wasita B, Mirawati DK, Budianto P, Suroto S. Tectona Grandis Leaves : Determination of Total Flavonoid Content , Phenolic Content , Characterization of the Leaves , and Compound Identification in GC-MS. 2023;15(1):1–6. DOI:10.5530/pj.2023.15.24
Xu B, Ganesan K, Mickymaray S, Alfaiz F, Thatchinamoorthi R, Aboody MS Al. Immunomodulatory and antineoplastic efficacy of common spices and their connection with phenolic antioxidants. 2020;3(2):15. DOI:10.31989/bchd.v3i2.687
Xiao H-Y, Li N, Duan JJ-W, Jiang B, Lu Z, Ngu K, et al. Biologic-like In Vivo Efficacy with Small Molecule Inhibitors of TNFα Identified Using Scaffold Hopping and Structure-Based Drug Design Approaches. J Med Chem. 2020 Dec;63(23):15050–15071. DOI:https://doi.org/10.1021/acs.jmedchem.0c01732
Parawansah, Rizki Rahmadi Pratama, Nuralifah, Arfan, Ruslin, Retno Widyowati S. Anti-rheumatoid arthritis potential of Acorus calamus L . extract by interleukin-17 inhibition : Molecular insights through an in silico study. J Pharm Pharmacogn Res. 2024;12(4):628–646. DOI:10.56499/jppres23.1818_12.4.628
Arfan A, Asnawi A, Aman LO. Marine Sponge Xestospongia sp.: A Promising Source for Tuberculosis Drug Development - Computational Insights into Mycobactin Biosynthesis Inhibition. Borneo J Pharm. 2024;7(1):40–50. DOI:https://doi.org/10.33084/bjop.v7i1.5513
Oleg T, Arthur J. O. AutoDock Vina: Improving the Speed and Accuracy of Docking with a New Scoring Function, Efficient Optimization, and Multithreading. J Comput Chem. 2010;31(2):455–461. DOI:https://doi.org/10.1002/jcc.21334
Arfan A, Muliadi R, Malina R, Trinovitasari N, Asnawi A. Docking and Dynamics Studies: Identifying the Binding Ability of Quercetin Analogs to the ADP-Ribose Phosphatase of SARS CoV-2. J Kartika Kim. 2022 Jan;5(2):145–151. DOI:https://doi.org/10.26874/jkk.v5i2.143
Lim HW, Kumar H, Kim BW, More SV, Kim IW, Park JI, et al. β-Asarone (cis-2,4,5-trimethoxy-1-allyl phenyl), attenuates pro-inflammatory mediators by inhibiting NF-κB signaling and the JNK pathway in LPS activated BV-2 microglia cells. Food Chem Toxicol. 2014;72:265–272. DOI:https://doi.org/10.1016/j.fct.2014.07.018
Wongwat T, Srihaphon K, Pitaksutheepong C, Boonyo W, Pitaksuteepong T. Suppression of inflammatory mediators and matrix metalloproteinase (MMP)-13 by Morus alba stem extract and oxyresveratrol in RAW 264.7 cells and C28/I2 human chondrocytes. J Tradit Complement Med. 2020;10(2):132–140. DOI:https://doi.org/10.1016/j.jtcme.2019.03.006
Gnanamani A. Effect of active fraction of methanolic extract of Acorus calamus on sterol metabolism of Candida albicans. 2008;8(1):243-250.
Robert M, Miossec P. IL-17 in rheumatoid arthritis and precision medicine: From synovitis expression to circulating bioactive levels. Front Med. 2019;5:364. DOI:10.3389/fmed.2018.00364
Cronstein BN, Aune TM. Methotrexate and its mechanisms of action in inflammatory arthritis. Nat Rev Rheumatol. 2020;16(3):145–154. DOI:https://doi.org/10.1038/s41584-020-0373-9
Liu X, Wang Z, Qian H, Tao W, Zhang Y, Hu C, et al. Natural medicines of targeted rheumatoid arthritis and its action mechanism. Front Immunol. 2022;13:945129. DOI:https://doi.org/10.3389/fimmu.2022.945129
Choy E. Understanding the dynamics: Pathways involved in the pathogenesis of rheumatoid arthritis. Rheumatol (United Kingdom). 2012;51(suppl_5):v3–1v1. DOI:https://doi.org/10.1093/rheumatology/kes113
Kondo N, Kuroda T, Kobayashi D. Cytokine Networks in the Pathogenesis of Rheumatoid Arthritis. International Journal of Molecular Sciences. 2021;22(20):10922. DOI:https://doi.org/10.3390/ijms222010922
Kim H, Han T-H, Lee S-G. Anti-inflammatory activity of a water extract of Acorus calamus L. leaves on keratinocyte HaCaT cells. J Ethnopharmacol. 2009;122(1):149–156. DOI:https://doi.org/10.1016/j.jep.2008.12.011
Sk MF, Jonniya NA, Roy R, Kar P. Unraveling the Molecular Mechanism of Recognition of Selected Next-Generation Antirheumatoid Arthritis Inhibitors by Janus Kinase 1. ACS Omega. 2022 Feb 22;7(7):6195–6209. DOI:https://doi.org/10.1021/acsomega.1c06715
Del Prado-Audelo ML, Cortés H, Caballero-Florán IH, González-Torres M, Escutia-Guadarrama L, Bernal-Chávez SA, Giraldo-Gomez DM, Magana JJ and Leyva-Gomez G. Therapeutic Applications of Terpenes on Inflammatory Diseases. Front Pharmacol. 2021;12:704197. DOI:https://doi.org/10.3389/fphar.2021.704197
Taams LS. Interleukin-17 in rheumatoid arthritis: Trials and tribulations. J Exp Med. 2020;217(3):e20192048. DOI:https://doi.org/10.1084/jem.20192048
Niu J, Cederstrand AJ, Eddinger GA, Yin B, Checco JW, Bingman CA, et al. Trimer-to-Monomer Disruption Mechanism for a Potent, Protease-Resistant Antagonist of Tumor Necrosis Factor-α Signaling. J Am Chem Soc. 2022 Jun;144(22):9610–9617. DOI: https://doi.org/10.1021/jacs.1c13717
Copyright (c) 2025 Jurnal Kefarmasian Indonesia

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.















